On a Self-Acting Apparatus for Multiplying and Maintaining Electric Charges, with Applications to Illustrate the Voltaic Theory
By Sir William Thomson (Lord Kelvin)
Proceedings of the Royal Society, June 20, 1867
[ED: Appearances of fractions altered from original due to HTML limitations]
401. In explaining the water-dropping collector for atmospheric electricity, in a lecture in the Royal Institute in 1860 (§ 285, above), I pointed out how, by disinsulating the water-jar and collecting the drops in an insulated vessel, a self-acting electric condenser is obtained. If, owing to electrified bodies in the neighbourhood, the potential in the air round the place where the stream breaks into drops is positive, the drops fall away negatively electrified; or vice versa, if the potential is negative, the drops fall away positively electrified. The stream of water descending does not in any way detract from the charges of the electrified bodies to which its electric action is due, provided always these bodies are kept properly insulated; but by the dynamical energy of fluid-motion, and work performed by gravity upon the descending drops, electricity may be unceasingly produced on the same principle as by the electrophorus. But, as in the electrophorus there was no provision except good insulation for maintaining the charge of the electrified body or bodies from which the induction originates, this want is supplied by the following reciprocal arrangement, in which the body charged by the drops of water in their turn keep up the charge of the inductor of the first.

Fig. 1.
402. To stems connected with the inside coatings of two Leyden phials are connected metal pieces, which, to avoid circumlocution, I shall call inductors and receivers. Each stem bears an inductor and a receiver, the inductor of the first jar being vertically over the receiver of the second jar, and vice versa. Each inductor consists of a vertical metal cylinder (fig. 1) open at each end. Each receiver consists of a vertical metal cylinder open at each end, but partially stopped in its middle by a small funnel (fig. 1), with its narrow mouth pointing downwards, and situated a little above the middle of the cylinder. Two fine vertical streams of uninsulated water are arranged to break into drops, one as near as may be to the centre of each inductor. The drops fall along the remainder of the axis of the inductor, and thence downwards, along the upper part of the axis of the receiver of the other jar, until they meet the funnel. The water re-forms into drops at the fine mouth of the funnel, which fall along the lower part of the axis of the receiver and are carried off by a proper drain below the apparatus. Suppose now a small positive charge of electricity be given to the first jar. Its inductor electrifies negatively each drop of water breaking away in its centre from the continuous uninsulated water above; all these drops give up their electricity to the second jar, when they meet the funnel in its receiver. The drops falling away from the lower fine mouth of the funnel carry away excessively little electricity, however highly the jar may be charged; because the place where they break away is, as it were, in the interior of a conductor, and therefore has nearly zero electrification. The negative electrification thus produced in the second jar acts, through its inductor, on the receiver of the first jar, and causes the negative electrification of the second jar to go on more rapidly, and so on. The dynamical value of the electrifications thus produced is drawn from the energy of the descending water, and is very approximately equal to the integral work done by gravity against electric force of the drops, in their path from the point where they break away from the uninsulated water above, to contact with the funnel of the receiver below. In the first part of this course each drop will be assisted downwards by electric repulsion from the inductively electrified water and tube above it; but below a certain point of its course the resultant electric force upon it will be upwards, and, according to the ordinary way of viewing the composition of electric forces, may be regarded as being at first chiefly upward repulsion of the receiver diminished by downward repulsion from the water and tube, and latterly the sum of upward repulsion of the receiver and upward attraction of the inductor. The potential method gives the integral amount, being the excess of work done against electric force, above work performed by electric force on each drop in its whole path. It is of course equal to m V, if m denote the quantity of electricity carried by each drop, as it breaks from the continuous water above, and V the potential of the inner coating of the jar bearing the receiver, the potential of the uninsulated water being taken as zero. The practical limit to the charges acquired is when one of them is so strong as to cause sparks to pass across some of the separating air-spaces, or to throw the drops of water out of their proper course and cause them to fall outside the receiver through which they ought to pass. It is curious, after commencing with no electricity except a feeble charge in one of the jars, only discoverable by a delicate electrometer, to see in the course of a few minutes a somewhat rapid succession of sparks pass in some part of the apparatus, or to see the drops of water scattered about over the lips of one or both the receivers.
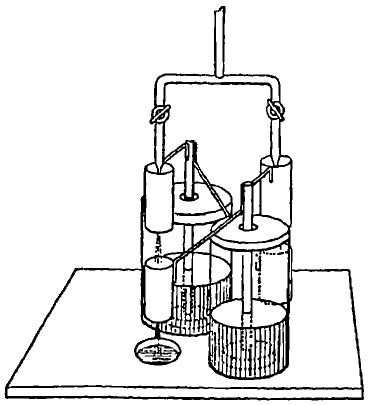
Fig. 2.
403. The Leyden jars represented in the sketch (fig. 2) are open-mouthed jars of ordinary flint glass, which, when very dry, I generally find to insulate electricity with wonderful perfection. The inside coatings consist of strong liquid sulphuric acid, and heavy lead tripods with vertical stems projecting upwards above the level of the acid, which, by arms projecting horizontally above the lip of the jar, bear the inductors and receivers, as shown in fig. 2. Lids of gutta percha or sheet metal close the mouth of each jar, except a small air-space of from 1/8 to 1/4 of an inch round the projecting stems. If a tube (fig. 3) be added to the lid to prevent currents of air from circulating into the interior of the jar, the insulation may be so good that the loss may be no more than one per cent. of the whole charge in three or four days. Two such jars may be kept permanently charged from year to year by very slow water-dropping arrangements, a drop from each nozzle once every two or three minutes being quite sufficient.

Fig. 3.
404. The mathematical theory of the action, appended below [1] , is particularly simple, but nevertheless curiously interesting.
405. The reciprocal electrostatic arrangement now described presents an interesting analogy to the self-sustaining electromagnetic systems recently brought before the Royal Society by Mr. C. W. Siemens and Professor Wheatstone, and mathematically investigated by Professor Clerk Maxwell. Indeed it was from the fundamental principle of this electromagnetic system that the reciprocal part of the electrostatic arrangement occurred to me recently. The particular form of self acting electrophorus condenser now described, I first constructed many years ago. I may take this opportunity of describing an application of it to illustrate a very important fundamental part of electric theory. I hope soon to communicate to the Royal Society a description of some other experiments which I made seven years ago on the same subject, and which I hope now to be able to prosecute further.
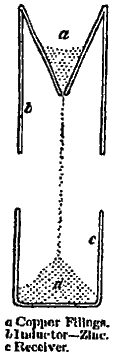
Fig. 4.
406. Using only a single inductor and a single receiver, as shown in fig. 1, let the inductor be put in metallic communication with a metal vessel of cistern whence the water flows; and let the receiver be put in communication with a delicate electroscope or electrometer. If the lining of the cistern and the inner metallic surface of the inductor be different metals, an electric effect if generally found to accumulate in the receiver and electrometer. Thus, for instance, if the inner surface of the inductor be dry polished zinc, and the vessel of water above be copper, the receiver acquires a continually increasing charge of negative electricity. There is little or no effect, either positive or negative, if the inductor present a surface of polished copper to the drops where they break from the continuous water above; but if the copper surface be oxidized by the heat of a lamp, until, instead of a bright metallic surface of copper, it presents a slate-coloured surface of oxide of copper to the drops, these become positively electrified, as is proved by a continually increasing positive charge exhibited by the electrometer. When the inner surface of the inductor is of bright metallic colour, either zinc or copper, there seems to be little difference in the effect whether it be wet with water or quite dry; also I have not found a considerable difference produced by lining the inner surface of the inductor with moist of dry paper. Copper filings falling from a copper funnel and breaking away from contact in the middle of a zinc inductor, in metallic communication with a copper funnel, as shown in fig. 4, produce a rapidly increasing negative charge in a small insulated can catching the below.
The quadrant divided-ring electrometer [2] indicating, by the image of a lamp on a scale, angular motions of a small concave mirror (1/3 of a grain in weight) such as I use in galvanometers, is very convenient for exhibiting these results. Its sensibility is such that it gives a deflection of 100 scale-divisions (1/40 of an inch each) on either side of zero, as the effect of a single cell of Daniell’s; the focusing, by small concave mirrors supplied to me by Mr. Becker, being so good that a deflection can easily be read with accuracy to a quarter of a scale-division. By adopting Peltier’s method of a small magnetic needle attached to the electric moveable body (or “needle”), and by using fixed steel magnets outside the instrument to give directing force (instead of the glass-fibre suspension of the divided-ring electrometers described in the articles referred to), and by giving a measurable motion by means of a micrometer screw to one of the quadrants, I have a few weeks ago succeeded in making this instrument into an independent electrometer, instead of a mere electroscope, or an electrometer in virtue of a separate gauge electrometer, as in the Kew recording atmospheric electrometer, described in the Royal Institution lecture.
407. Reverting to the arrangement described above of a copper vessel of water discharging water in drops from a nozzle through an inductor of zinc in metallic connection with the copper, let the receiver be connected with a second inductor, this inductor insulated; and let a second nozzle, from an uninsulated stream of water, discharge drops through it to a second receiver. Let this second receiver be connected with a inductor used to electrify a third stream of water to be caught in a third receiver, and so on. We thus have an ascending scale of electrophorus action analogous to the beautiful mechanical electric multiplier of Mr. C. F. Varley, with which, by purely electrostatic induction, he obtained a rapid succession of sparks from an ordinary single voltaic element. This result is easily obtained by the self-acting arrangement now described, with the important modification in the voltaic element according to which no chemical action is called into play, and work done by gravity is substituted for work done by the combination of chemical elements.
Footnotes
[ED: Equation appearances altered from original due to HTML limitations. Also, the print in the copy used was much reduced so there may be errors; if there’s any doubt, please consult original source for accurate equations before you electrocute yourself.]
-
[1] Let c, c′ be the capacities of the two jars, l, l′ their rates of loss per unit potential of charge, per unit of time, and D, D′ the values of the water-droppers influenced by them. Let +v and -v′ be their potentials at time t; v and v′ being of one sign in the ordinary use of the apparatus described in the text. The action is expressed by the following equations:—
c ( dv / dt ) = D′v′ - lv;
c′ ( dv′ / dt ) = Dv - l′v′.If c, D, l, c′, D′, l′ were all constant, the solution of these equations would be for the case of commencing with the first jar charged to potential 1, and the second zero,
v = ( ( c′ρ + l′ ) ερt - ( c′σ + l′ ) εσt ) / ( c′ ( ρ - σ ) ),
v′ = D ( ( ερt - εσt ) / ( c′ ( ρ - σ ) ) ),With the corresponding symmetrical expression for the case in which the second jar is charged, and the first at zero, in the beginning; the roots of the quadratic
( cx + l ) ( c′x + l′ ) - DD′ = 0
being denoted by ρ and σ. When ll′ > DD′, both roots are negative; and the electrification comes to zero in time, whatever may be the initial charges. But when ll′ < DD′, one root is positive and the other negative, and ultimately the charges augment in proportion to ερt if ρ be the positive root.
-
[2] See Nichol’s Encyclopædia, 1860, article “Electricity, Atmospheric;” or Proceedings of the Royal Institution, May 1860, Lecture on Atmospheric Electricity [§§ 249...293, above].